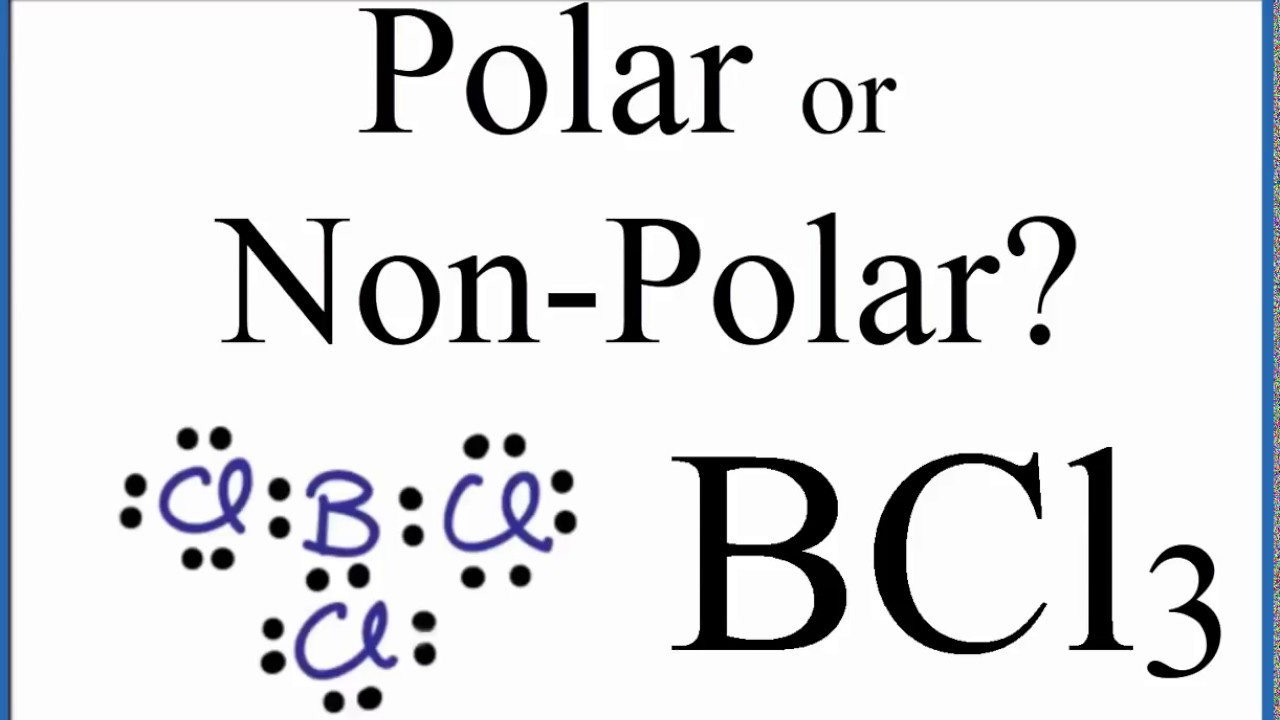BCl3 Lewis Structure (Boron Trichloride) | BCl3 Lewis Structure (Boron Trichloride) Welcome back to our channel and in today's video we will help you determine the Lewis Structure of Boron... | By

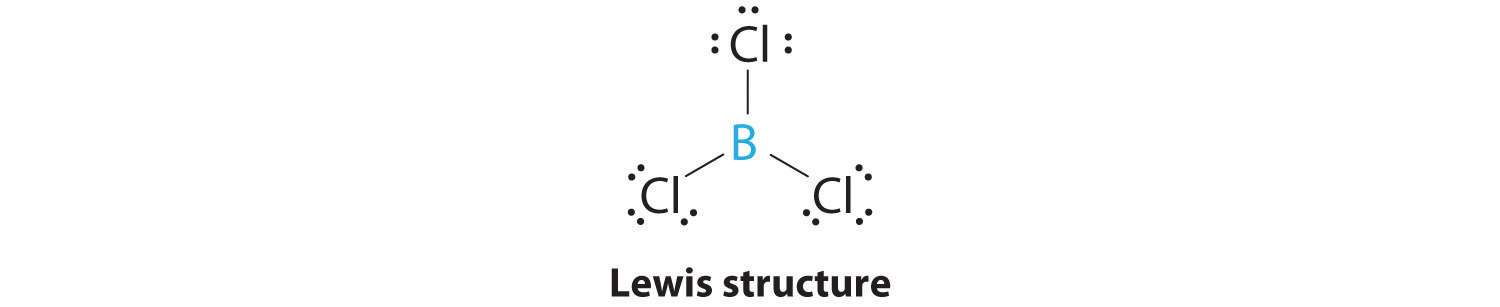
SOLVED: How many valence electrons does element 15 have? d. 2 Boron trichloride (BCl3) violates the octet rule: How many electrons are on the boron atom in BCl3? Which one of the

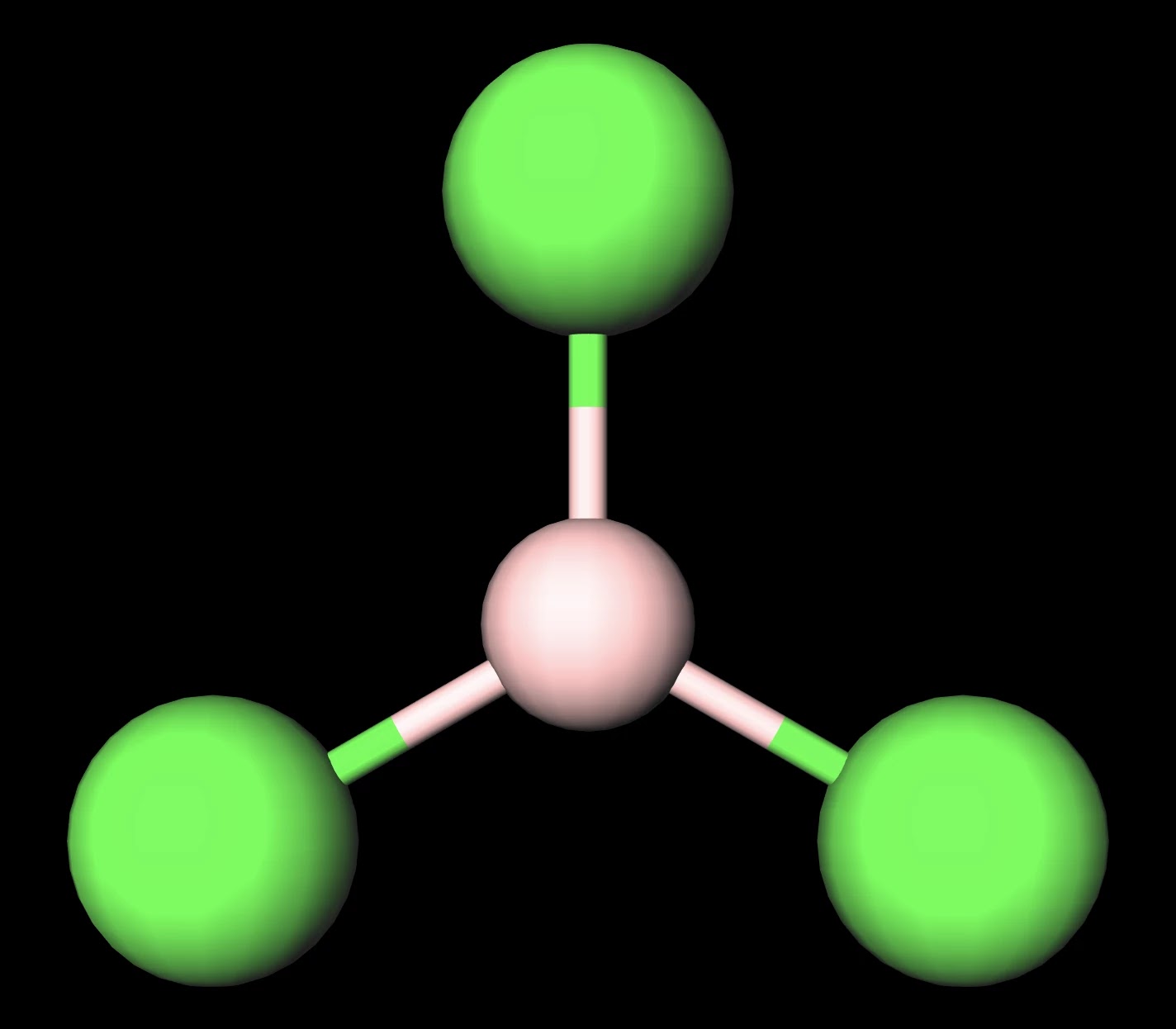
Why is BCl3 trigonal planar in shape while anhydrous AlCl3 is tetrahedral - Chemistry - Chemical Bonding and Molecular Structure - 10436299 | Meritnation.com

BCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle | Molecular geometry, Molecular, Vsepr theory

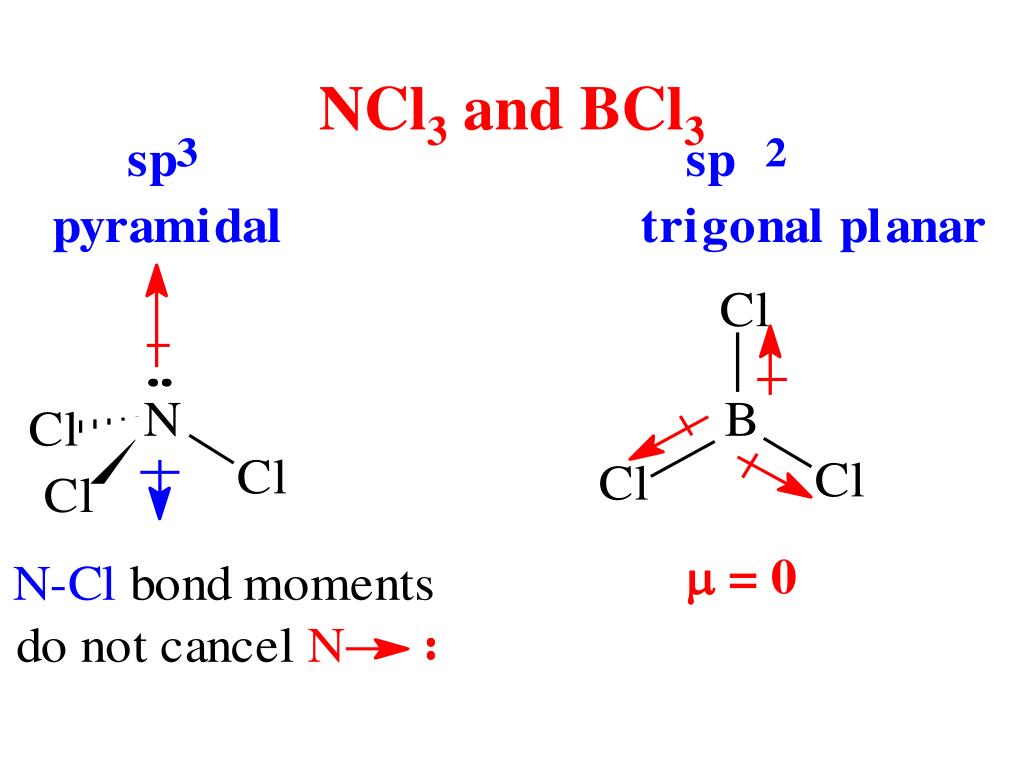
SOLVED: Molecular Structure: The VSEPR Model Examples: Moment of Eq = Predict shape and polarity A. polar (μ ≠0) B. nonpolar (μ = 0) PCl3 Vs BCl3 PCl5 Vs SbCl5 SF4
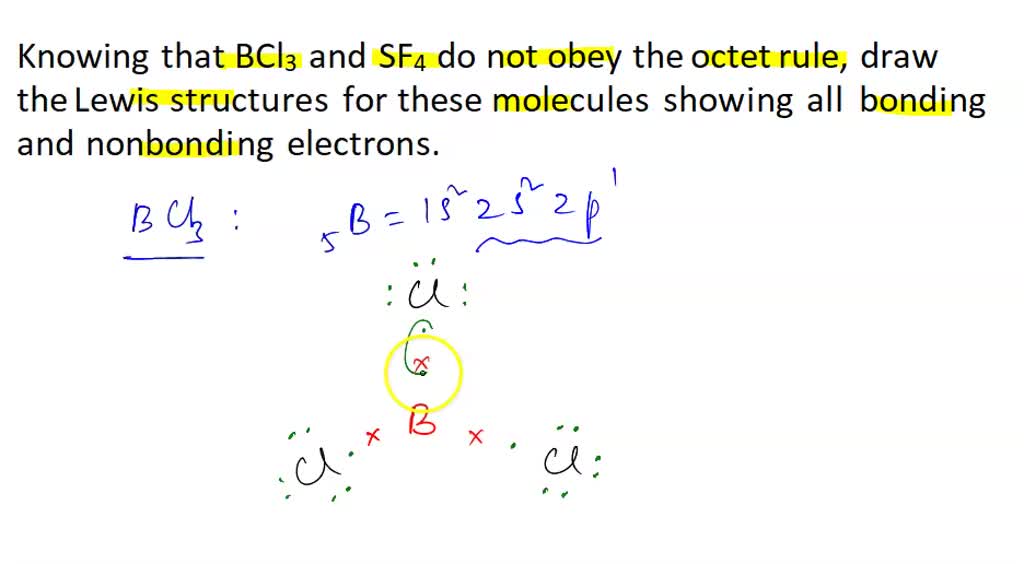
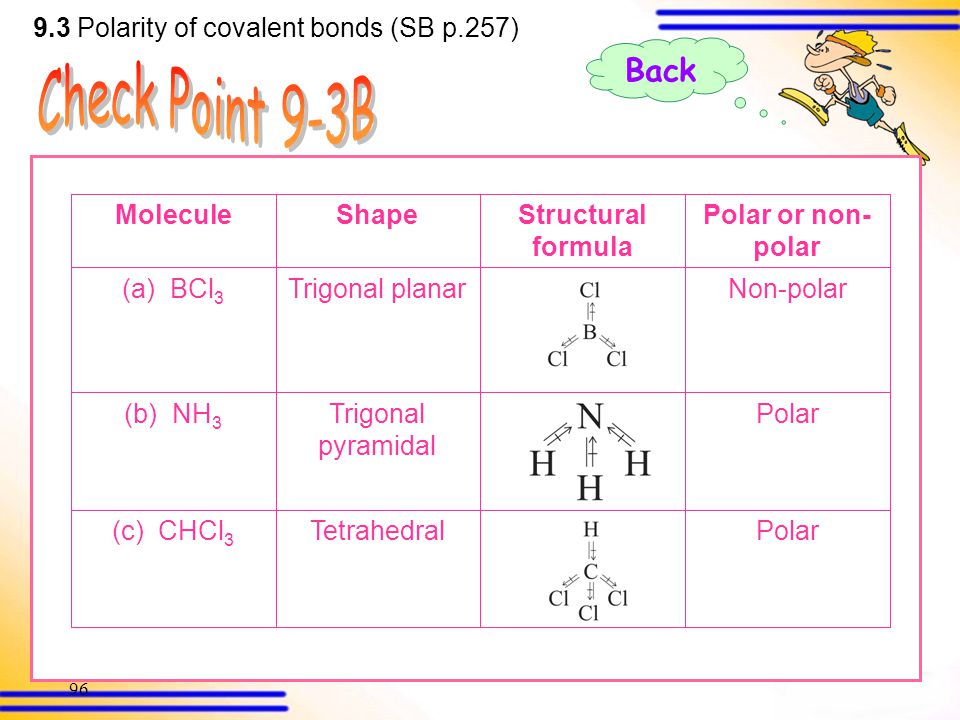
SOLVED: 1) Knowing that BCl3 and SF4 do not obey the octet rule, draw the Lewis structures for these molecules, showing all bonding and nonbonding electrons. 2) Draw Lewis structures for NH3,