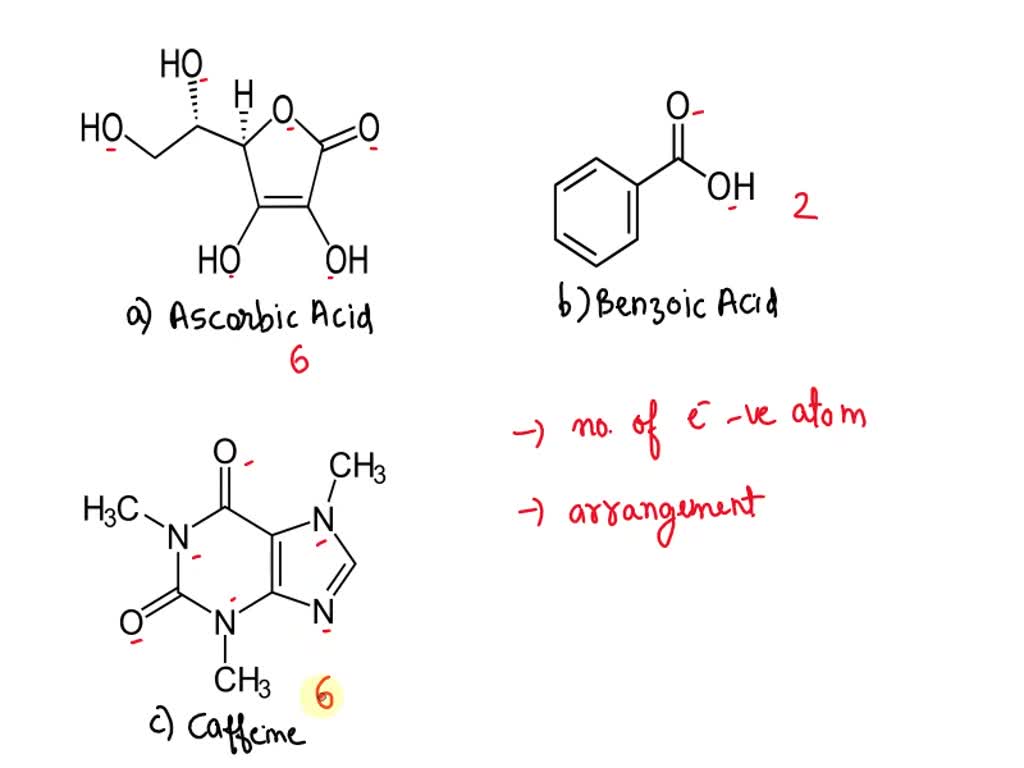
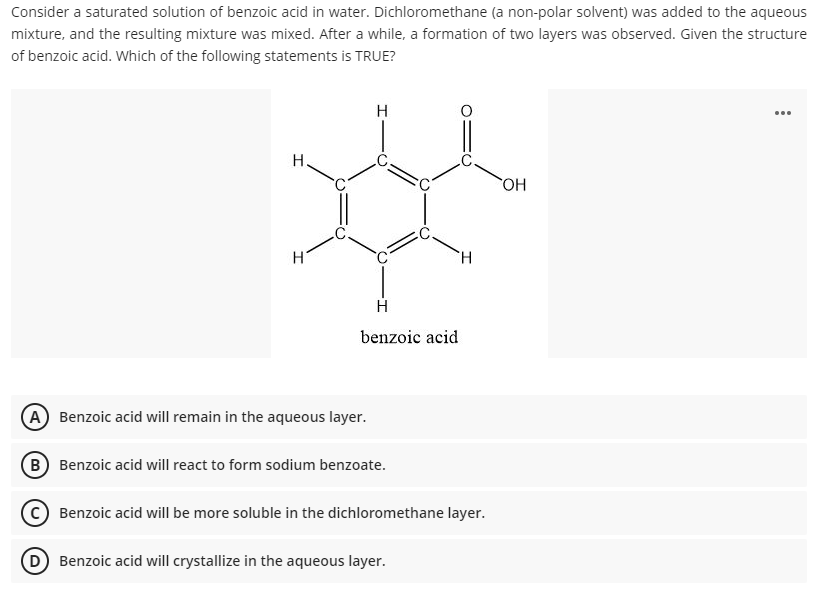
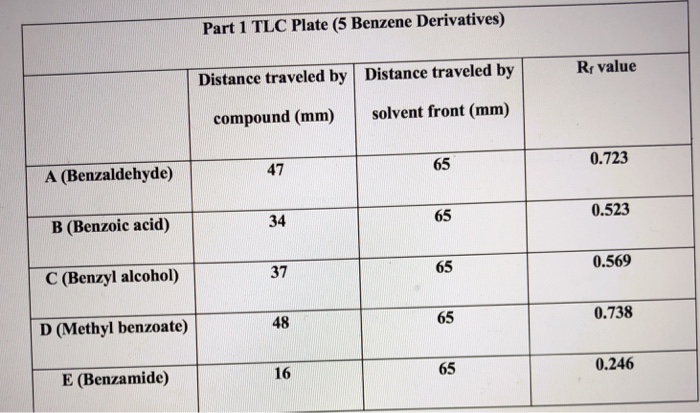
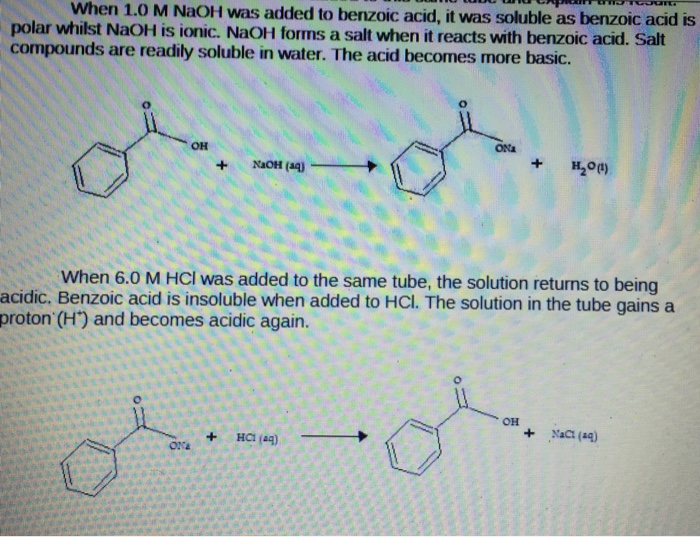
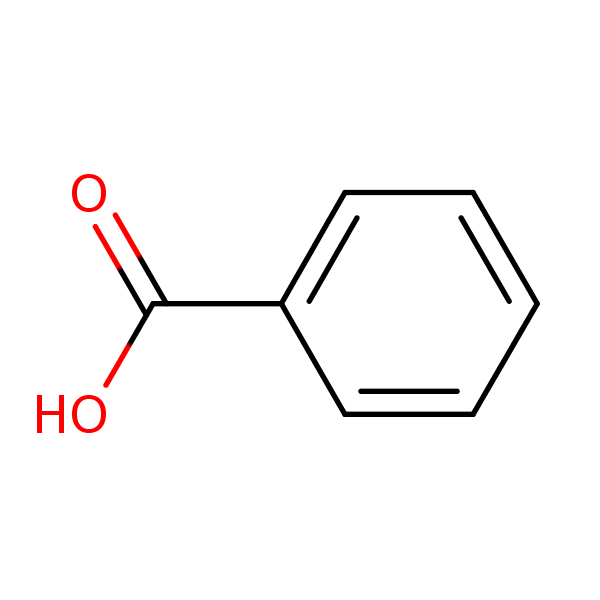
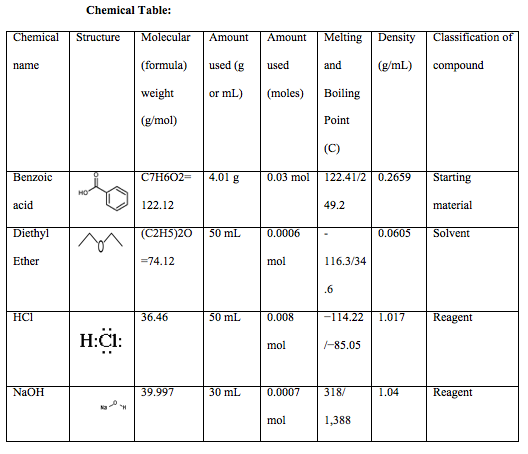
2. Draw the chemical structure of benzoic acid and use it to answer the following solubility-based questions: a) Circle and label the region of the molecule that exhibits non-polar behavior. b) Thes

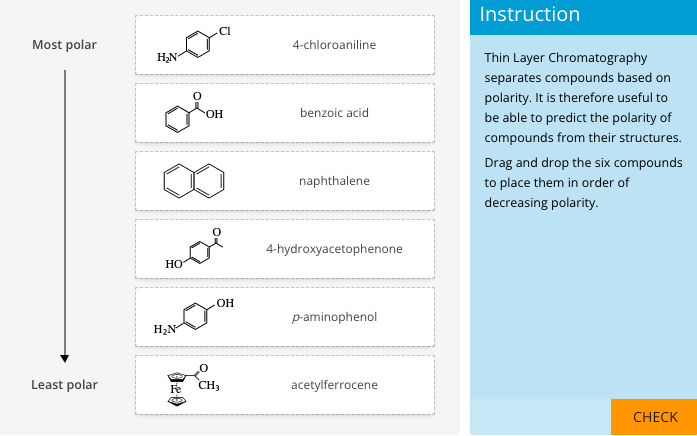
Examine the structures of the following functionalized compounds and then rank in order of increasing elution sequence: benzoic acid, acetophenone, benzyl alcohol, benzylamine, benzylamine hydrochlori | Homework.Study.com
How was a mixture of organic molecules (phenol, benzoic acid, p-toluidine and anisole) separated and isolating process? - Quora

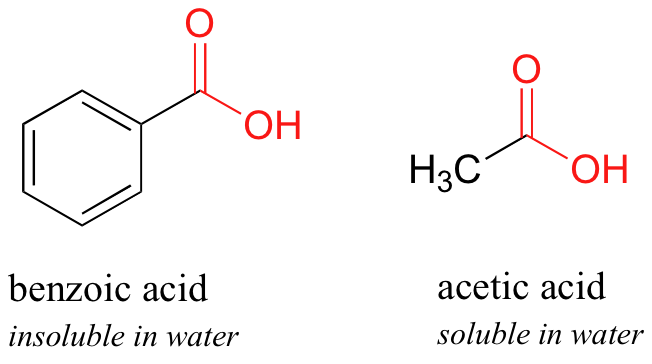
How can benzoic acid dissolve into diethyl ether since benzoic acid is polar and diethyl ether is non-polar? | Homework.Study.com

1) Rank trans-stilbene, benzoic acid, and fluorenone by polarity (least polar to most polar). Provide an explanation for your proposed ranking. (2) Hexane, ethyl acetate and acetone will be used as
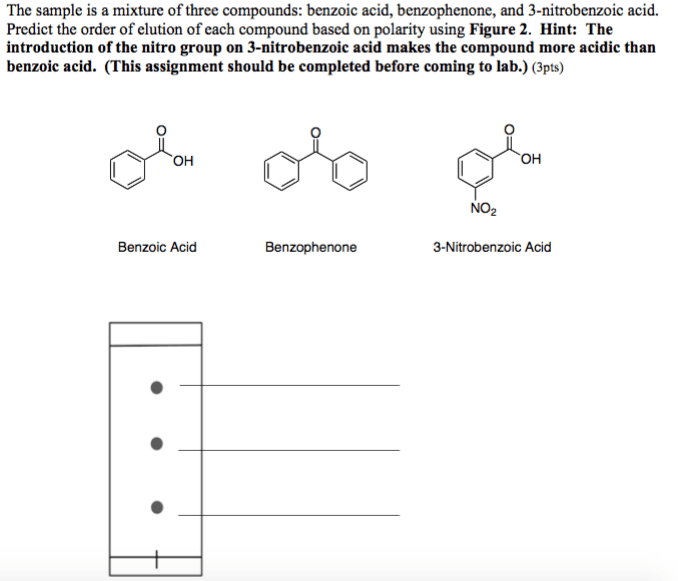
OneClass: The sample is a mixture of three compounds: benzoic acid, benzophenone, and 3-nitrobenzoic ...
Between acetylsalicylic acid and benzoic acid, which is most polar if you look only at the structure (not at solubility in water g/l), and why? Why is that structure most polar? -