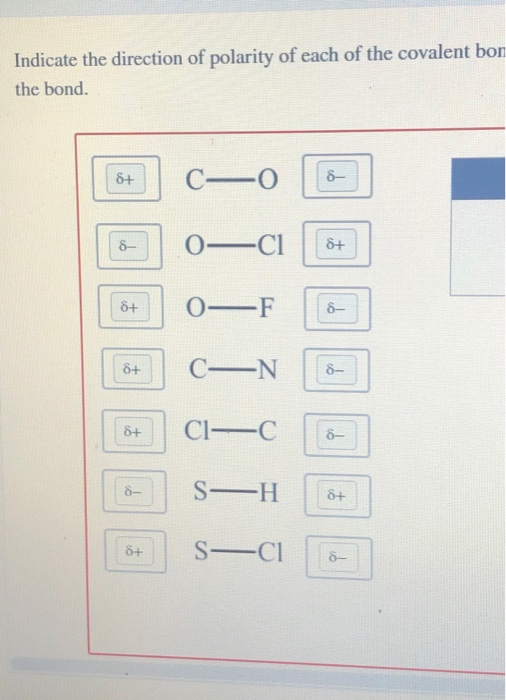
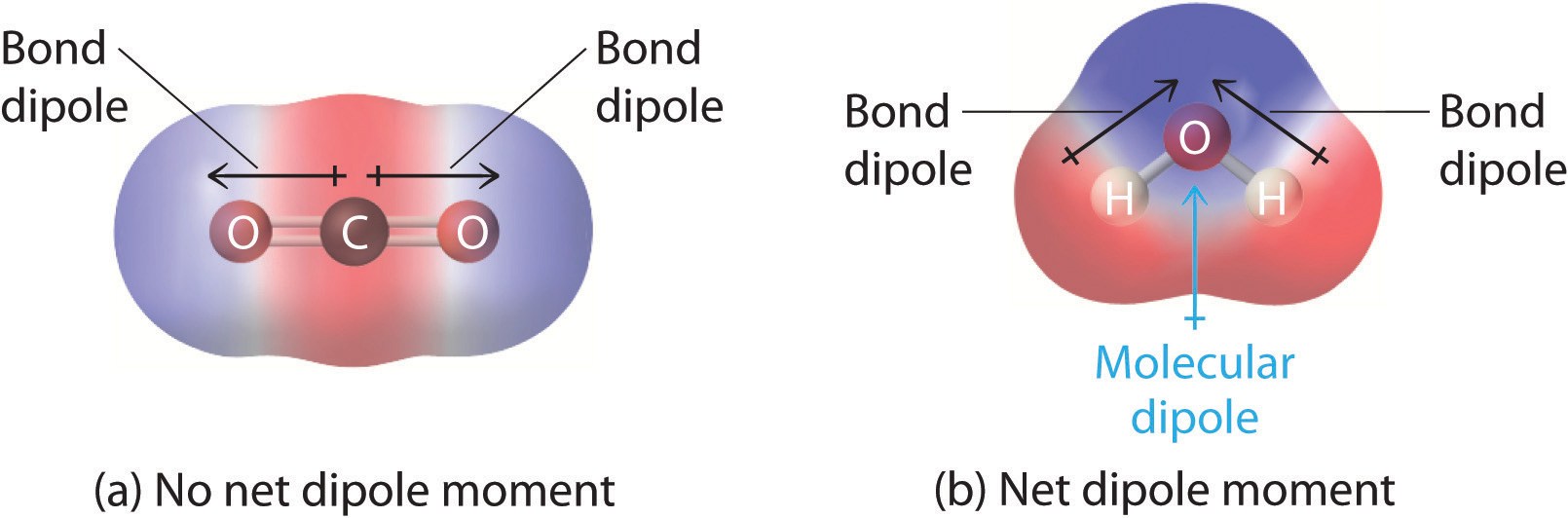
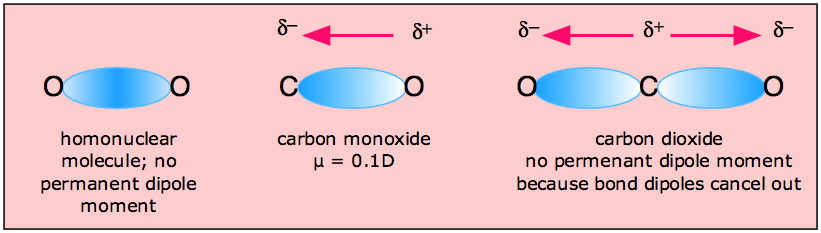
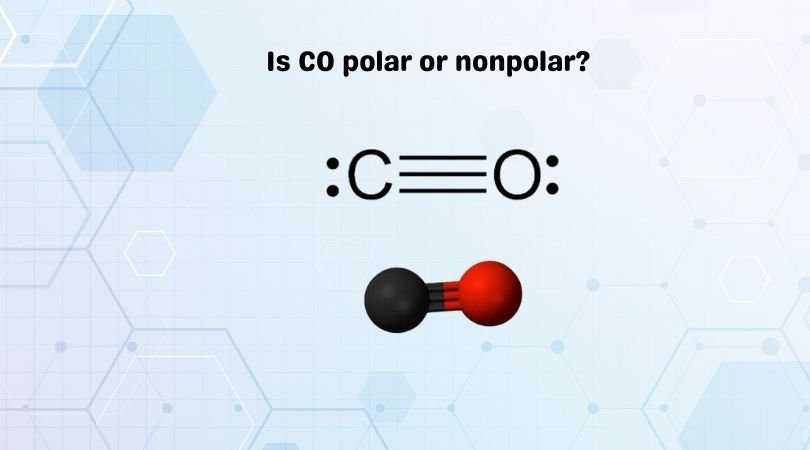
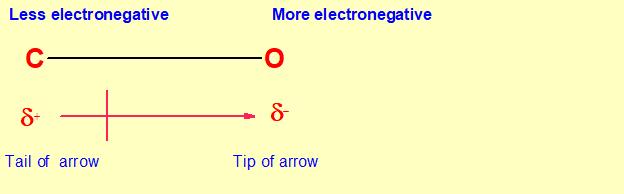
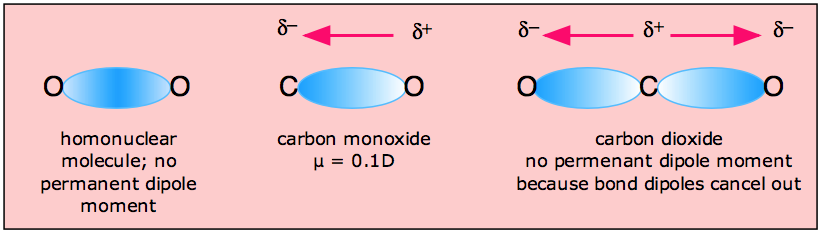
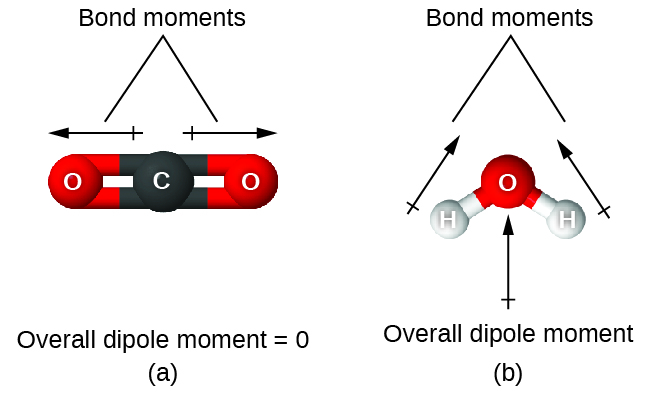
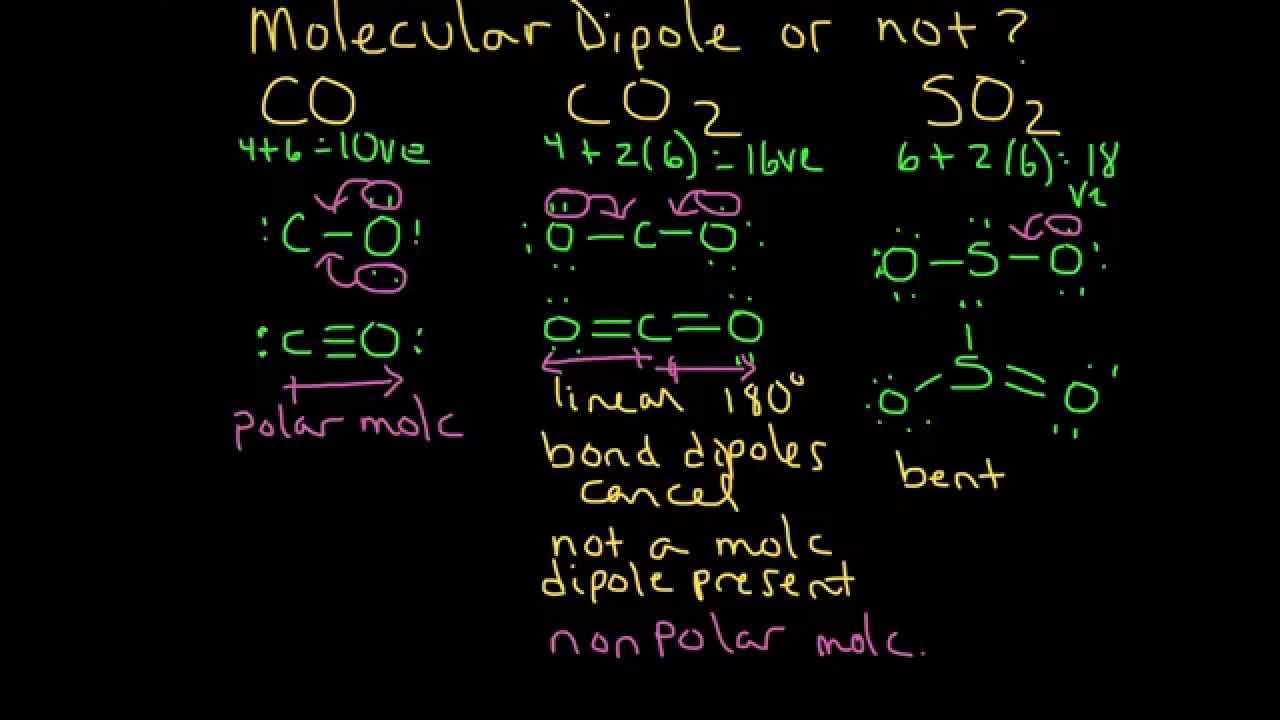How does the polarity of C O bond increases with increase in alkyl geoups attached to the Carbon atom containing OH group?
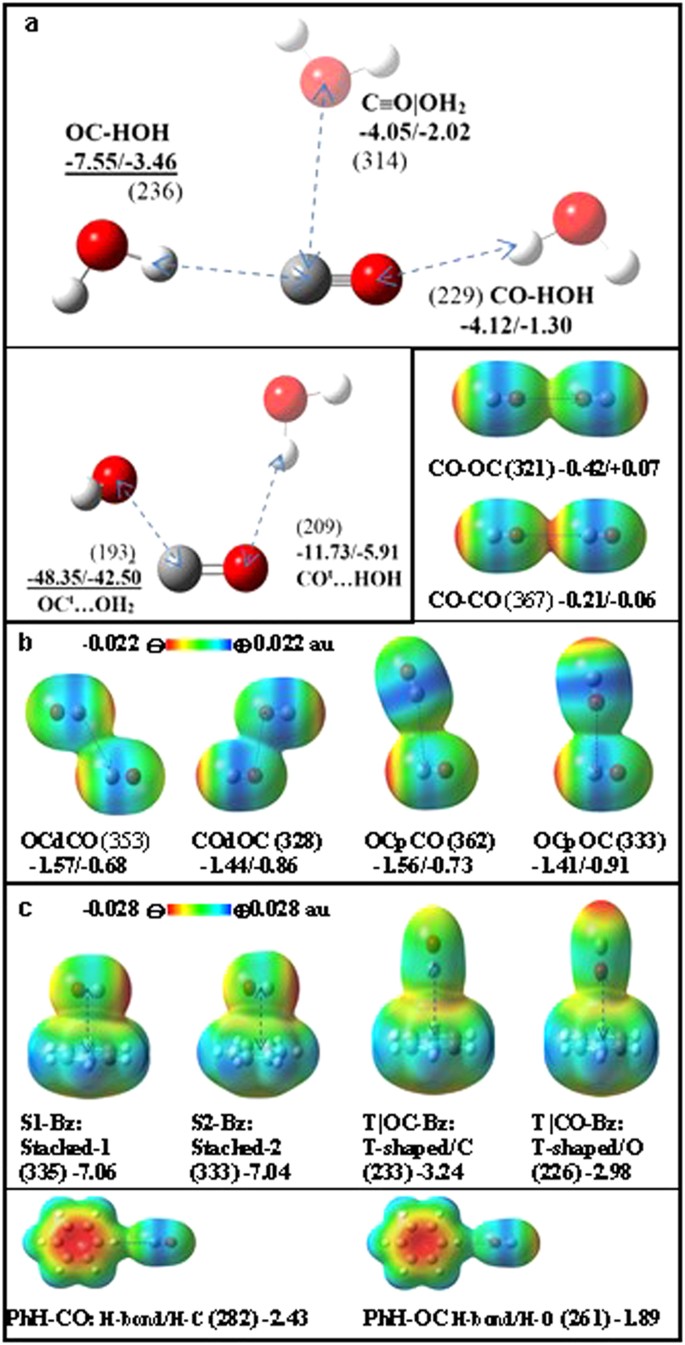
Intriguing Electrostatic Potential of CO: Negative Bond-ends and Positive Bond-cylindrical-surface | Scientific Reports

Must polar bonds give rise to polar molecules? And, why is water a polar molecule? | anhourofchemaday
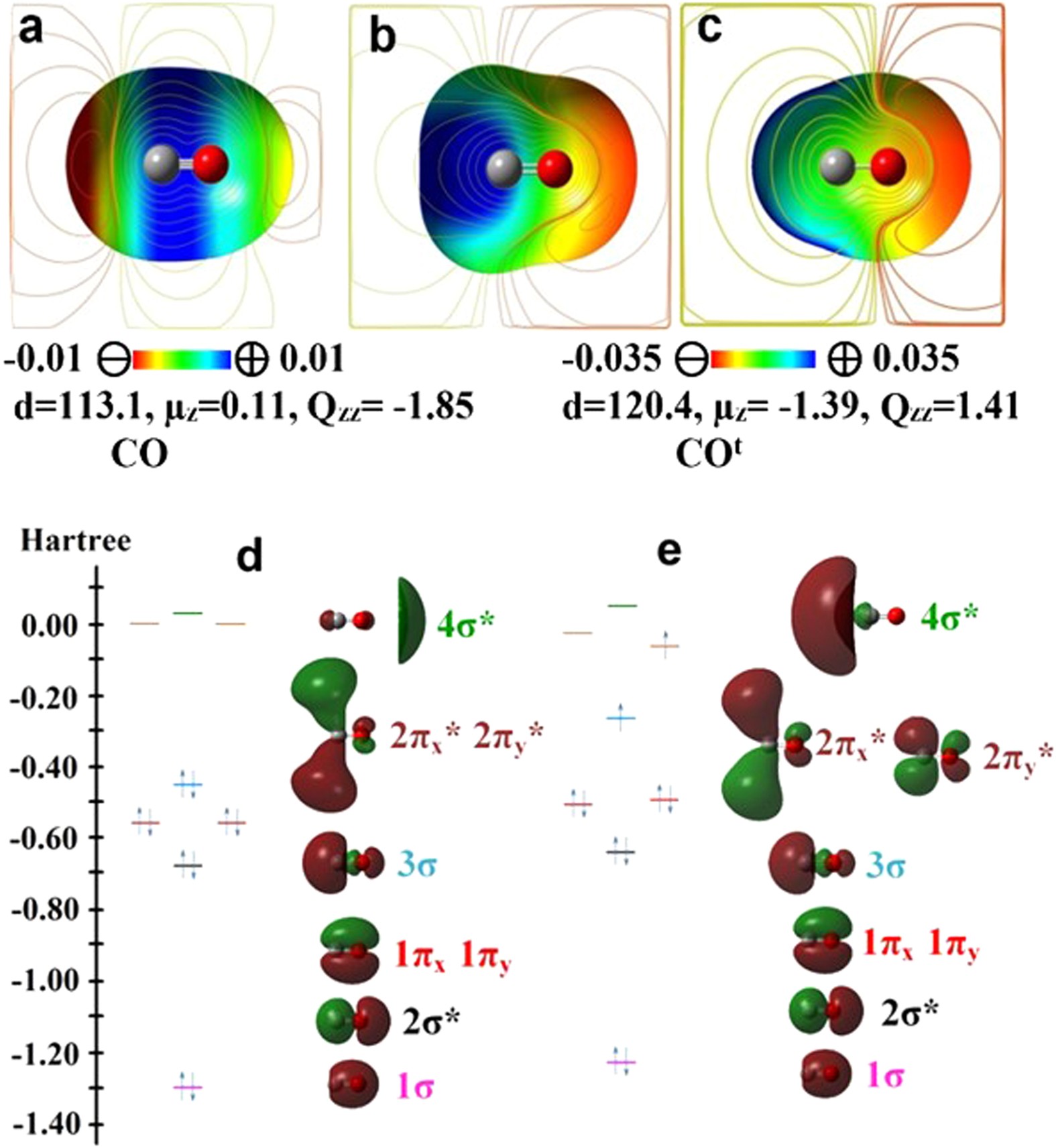
Intriguing Electrostatic Potential of CO: Negative Bond-ends and Positive Bond-cylindrical-surface | Scientific Reports