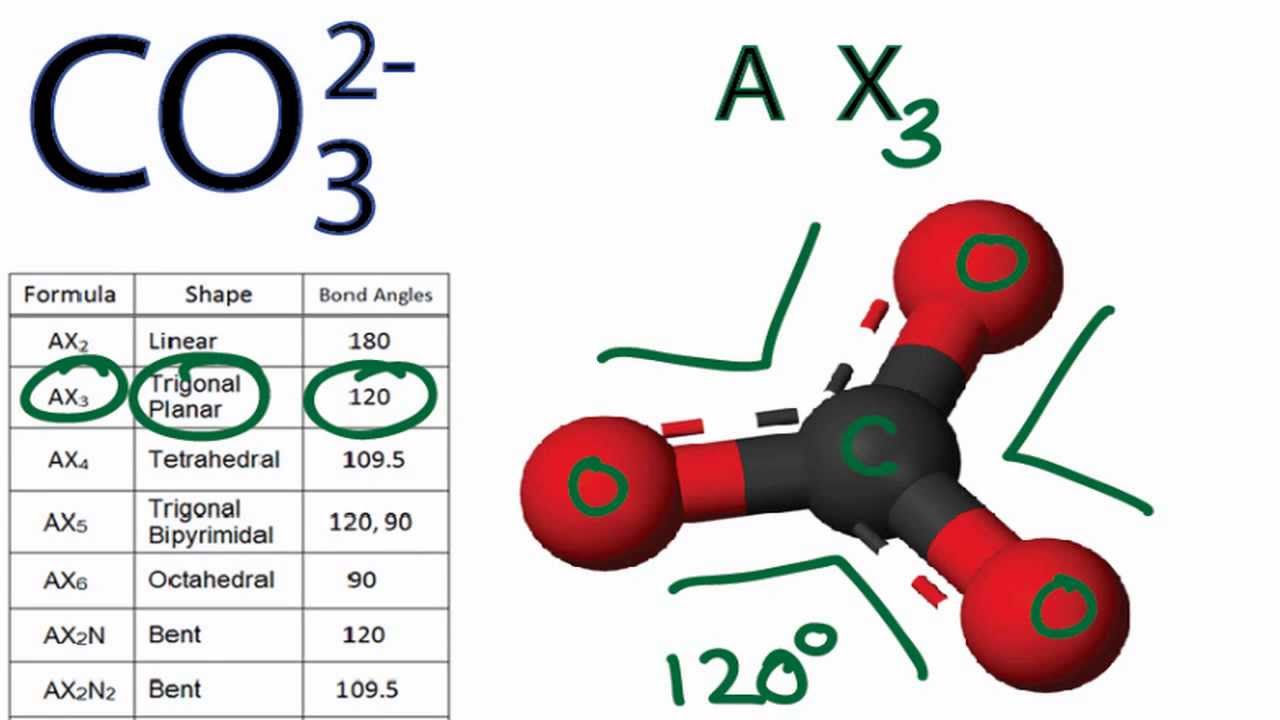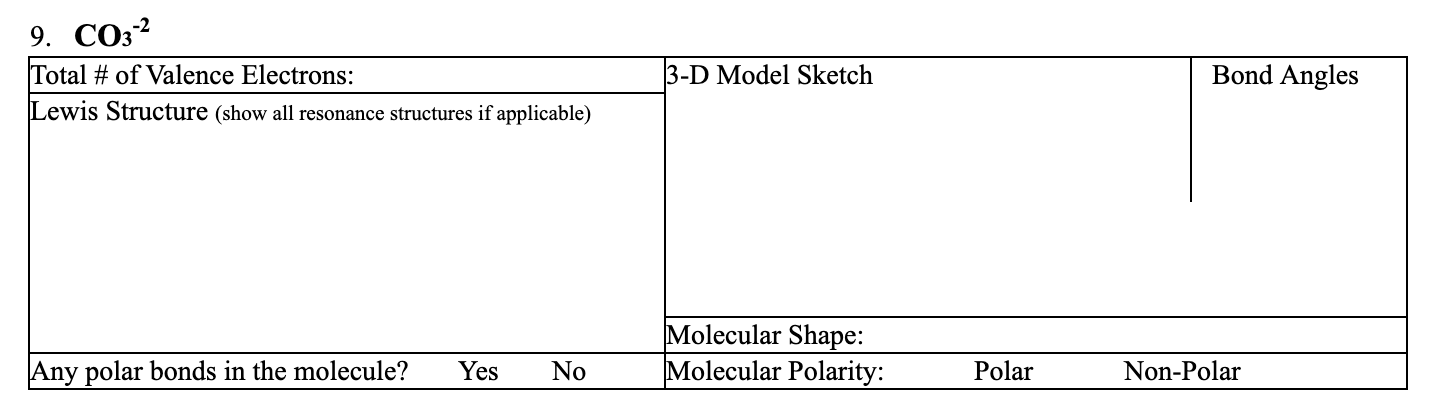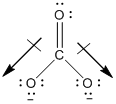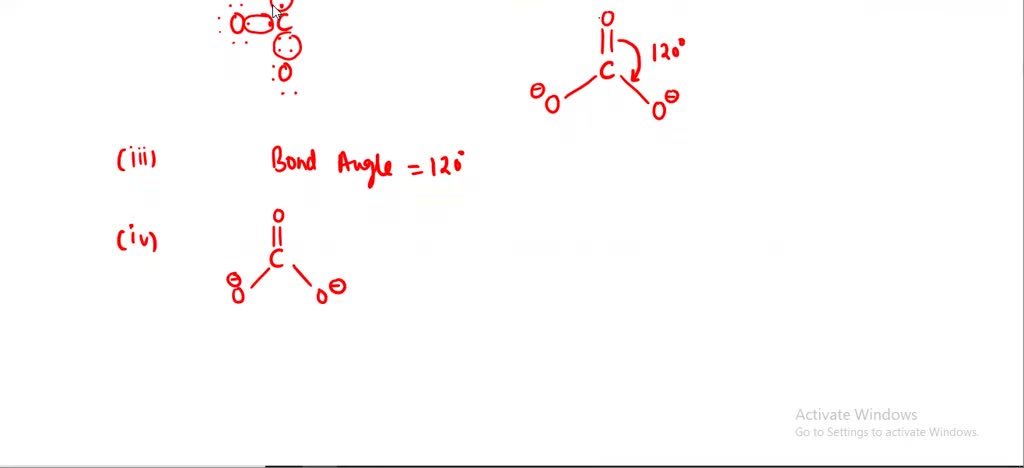
SOLVED: Draw the lewis structure of CO3^2- 1. give the predicted structure for CO3^2- based on VSEPR 2. give the hybridization for the central atom in CO3^2- 3. give the bond angle(s)
amongst NO3^ 1, AsO3^ 3,CO3^ 2, ClO3^ 1, SO3^ 2 and BO3^ 3, the non polar species are: (1) NO3^ 1,CO3^ 2, BO3^ 3 (2) AsO3^ 3, ClO3^ 1, SO3^ 2 (3) CO3^ 2, SO3^ 2, AsO3 3 (4) ClO3^ 1, SO3^ 2, NO3^ 1

Determine if each compound or ion below has a dipole moment. (a) Carbonate ion (CO_3 ^{2-}) | Homework.Study.com