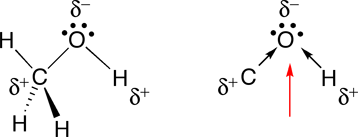Chemical polarity Methanol Covalent bond Lewis structure Molecule, induced, angle, white png | PNGEgg

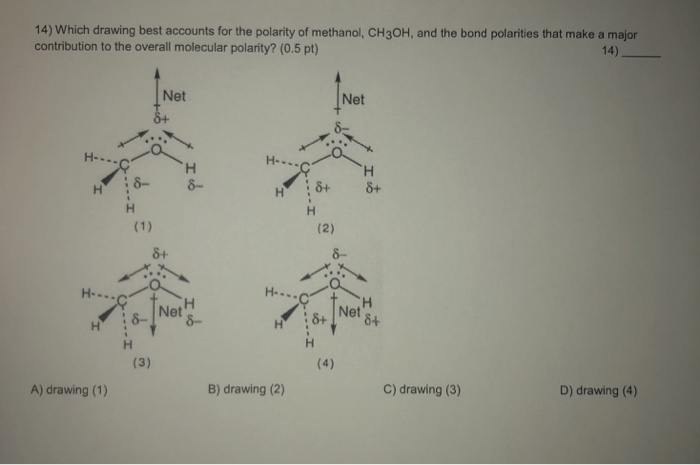
Which drawing best accounts for the polarity of methanol, CH3OH, and the bond polarities that make a major - brainly.com
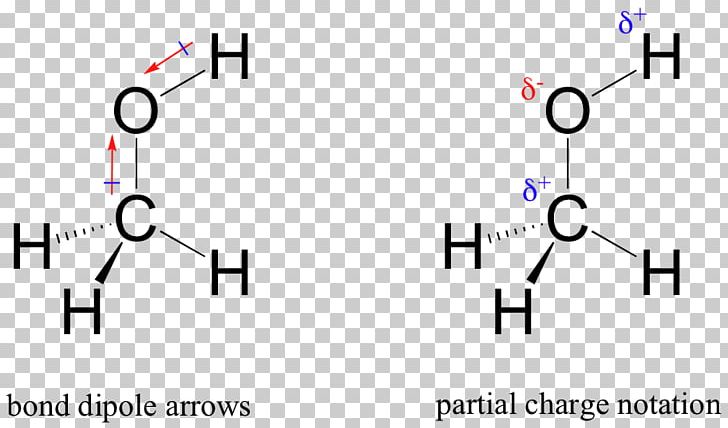
Chemical Polarity Methanol Covalent Bond Lewis Structure Molecule PNG, Clipart, Angle, Apolaire Verbinding, Bond Dipole Moment,

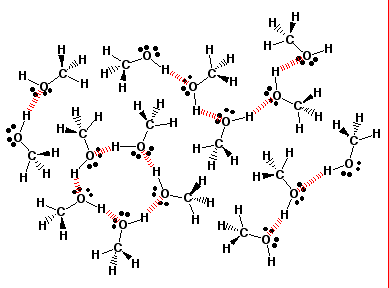
SOLVED: Liquid methanol CH3OH 32.04 g/mol Lewis Structure: (consider VSEPR shape - mark-up label the structure to help you) Intermolecular Force (IMF) Identification: Identify what IMF's are present in sample many molecules (

What characteristics do methanol, chloroform, diethyl ether, etc. possess that makes them good solvents? - Quora




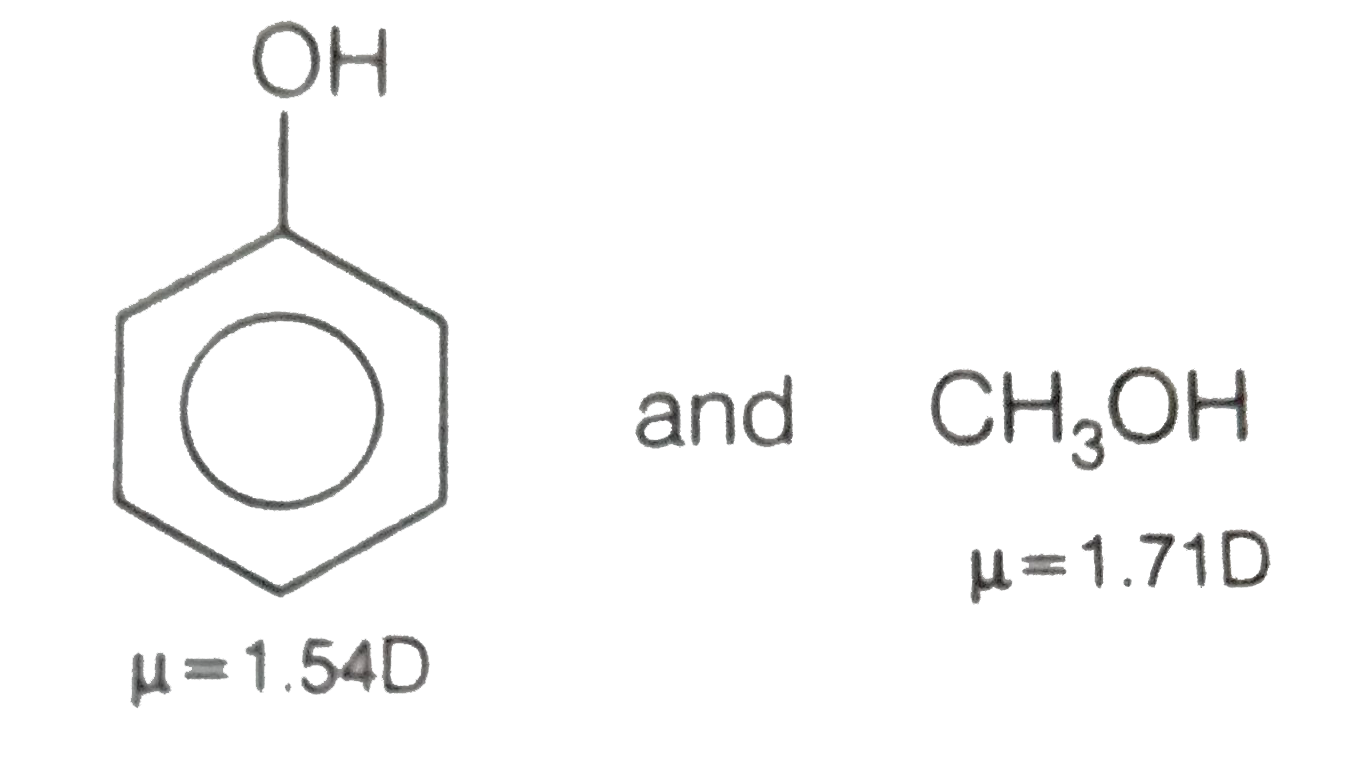

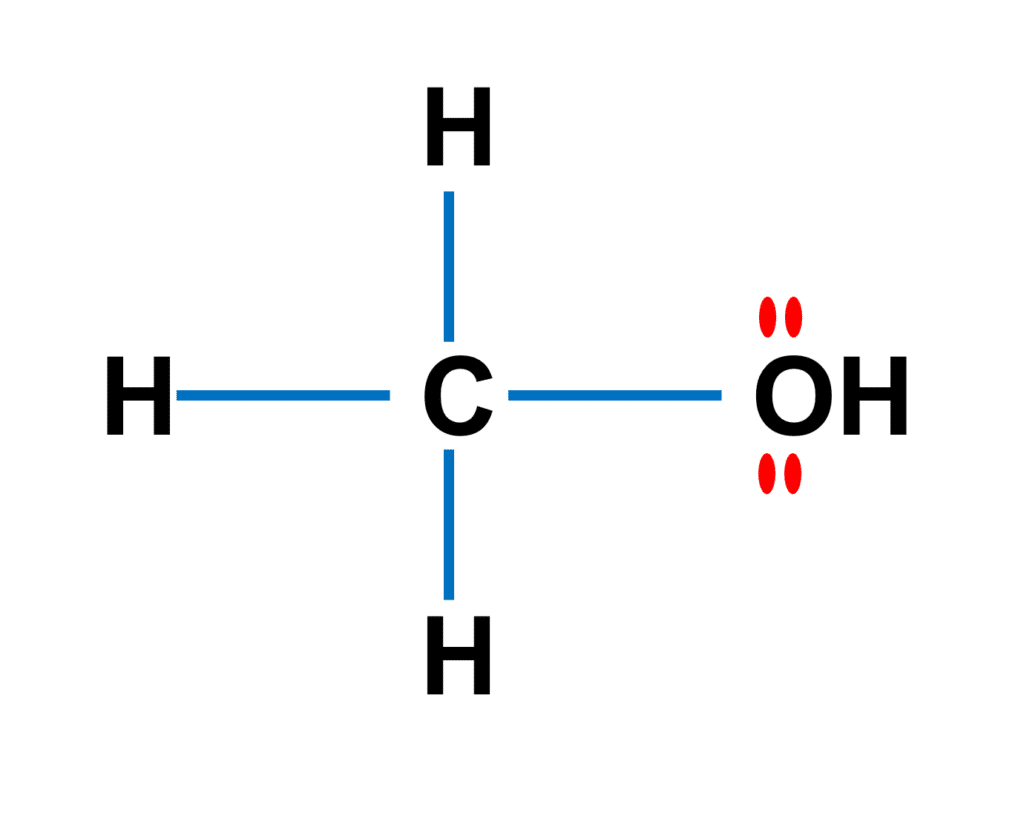
![Is the \\[O - H\\] bond in methanol polar or nonpolar? Is the \\[O - H\\] bond in methanol polar or nonpolar?](https://www.vedantu.com/question-sets/85482c56-aa98-4c21-899b-3c34c5fa0cca7552265523646111858.png)