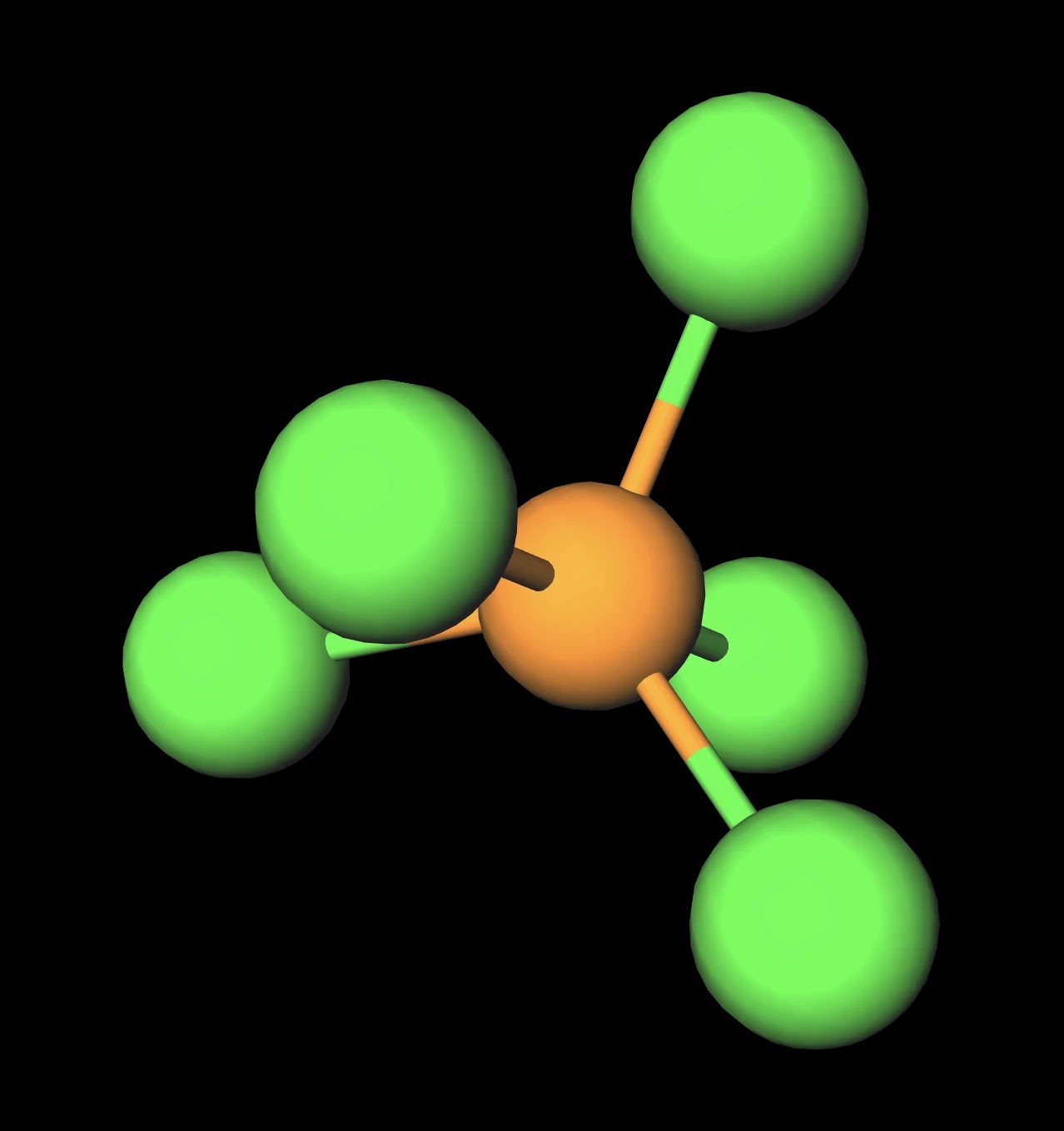
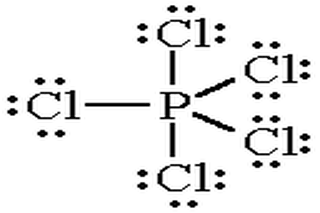
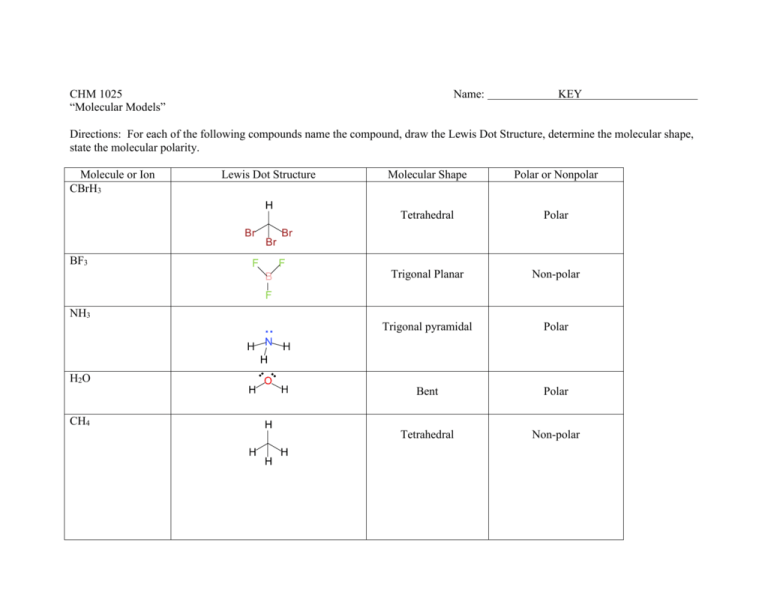
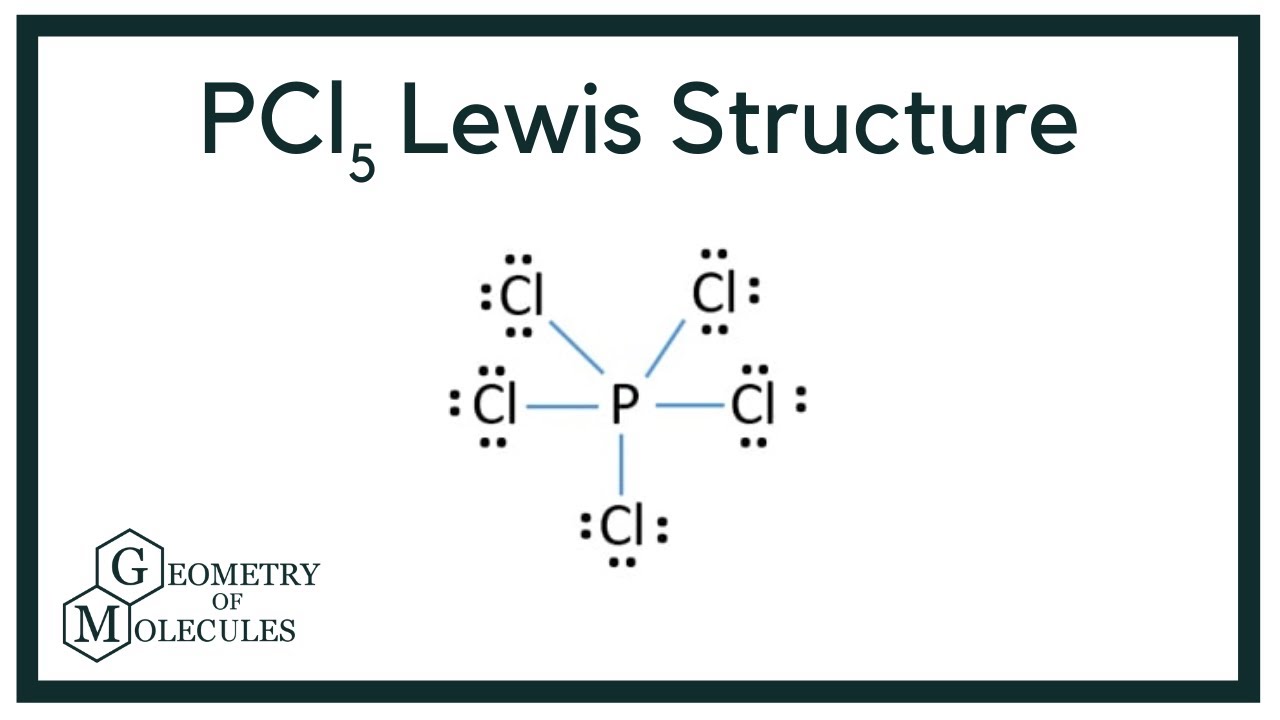
SOLVED: Molecular Structure: The VSEPR Model Examples: Moment of Eq = Predict shape and polarity A. polar (μ ≠0) B. nonpolar (μ = 0) PCl3 Vs BCl3 PCl5 Vs SbCl5 SF4

Which solvent, water or carbon tetrachloride, would you choose to dissolve each of the following? a. KrF_2 b. SF_2 c. SO_2 d. CO_2 e. MgF_2 f. CH_2O g. CH_2=CH_2 | Homework.Study.com















![IMAT 2018 Q47 [Polarity] - IMAT Past Papers Solutions - IMAT 2023 IMAT 2018 Q47 [Polarity] - IMAT Past Papers Solutions - IMAT 2023](https://imat.entermedschool.com/uploads/default/original/2X/8/865d4315f934a57068f643ee7b954c18b2ec789e.png)