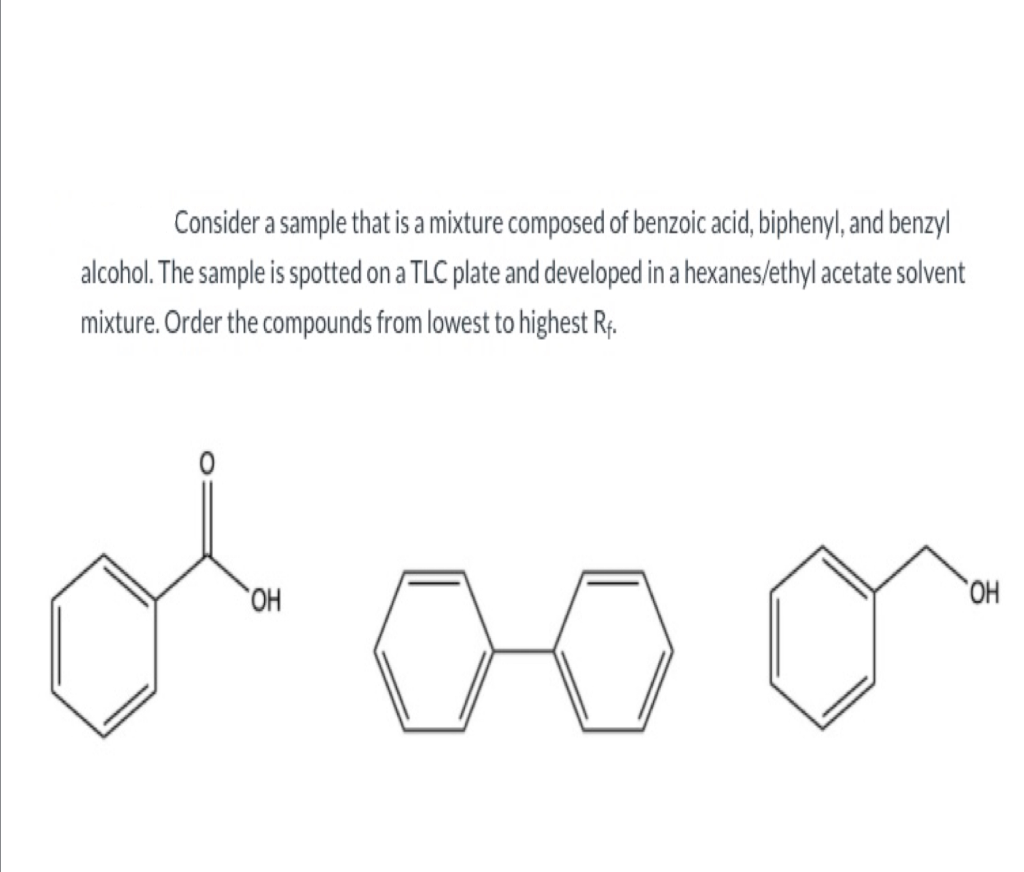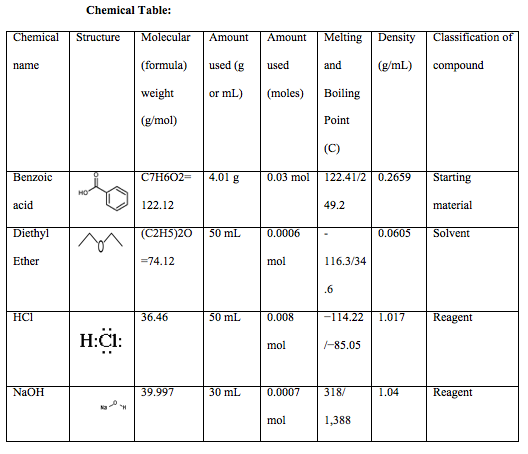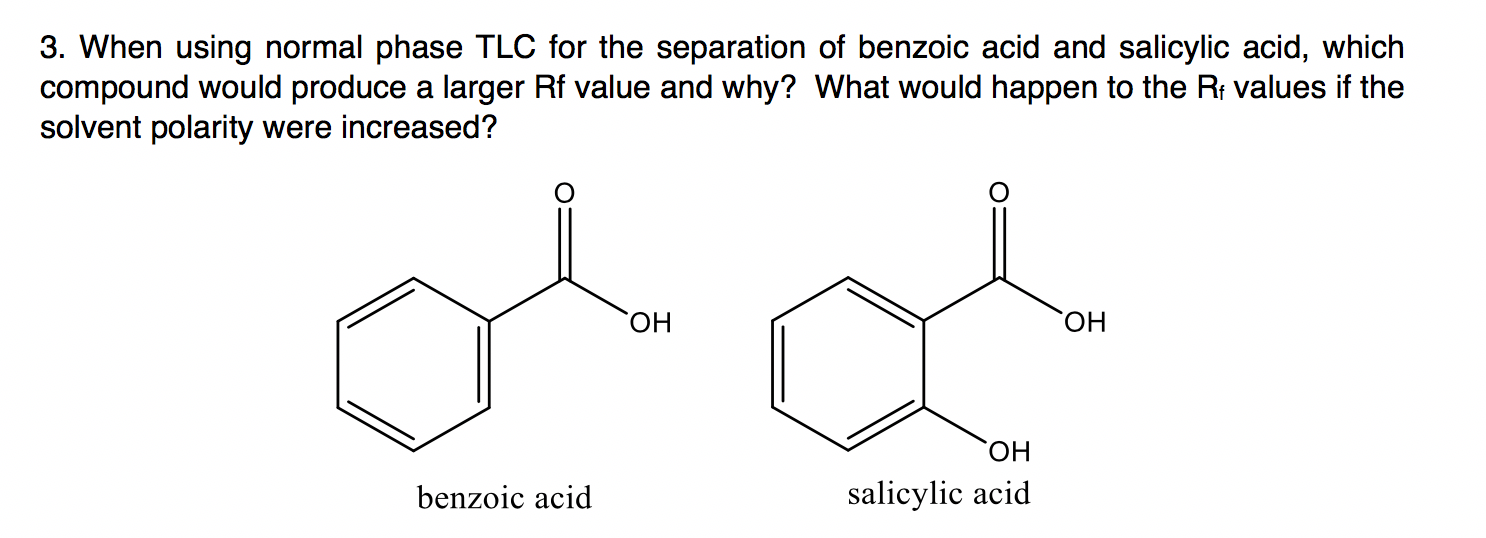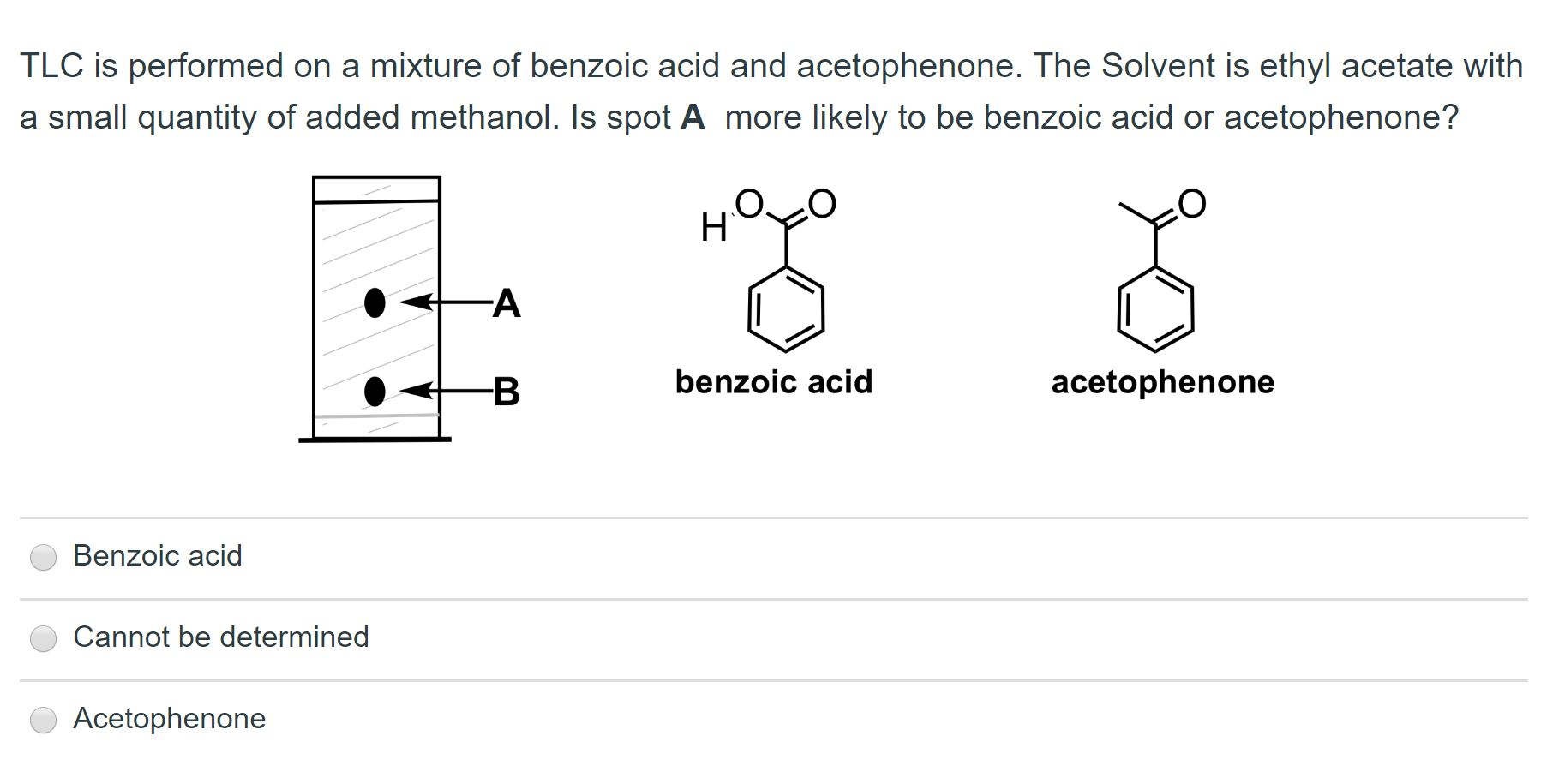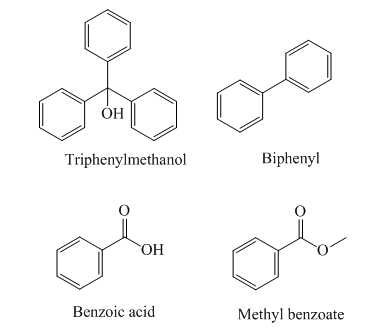
The order of elution of a mixture of triphenylmethanol, biphenyl, benzoic acid, and methyl benzoate from an alumina column should be predicted. Concept introduction: A separation technique used for the segregation of
How was a mixture of organic molecules (phenol, benzoic acid, p-toluidine and anisole) separated and isolating process? - Quora
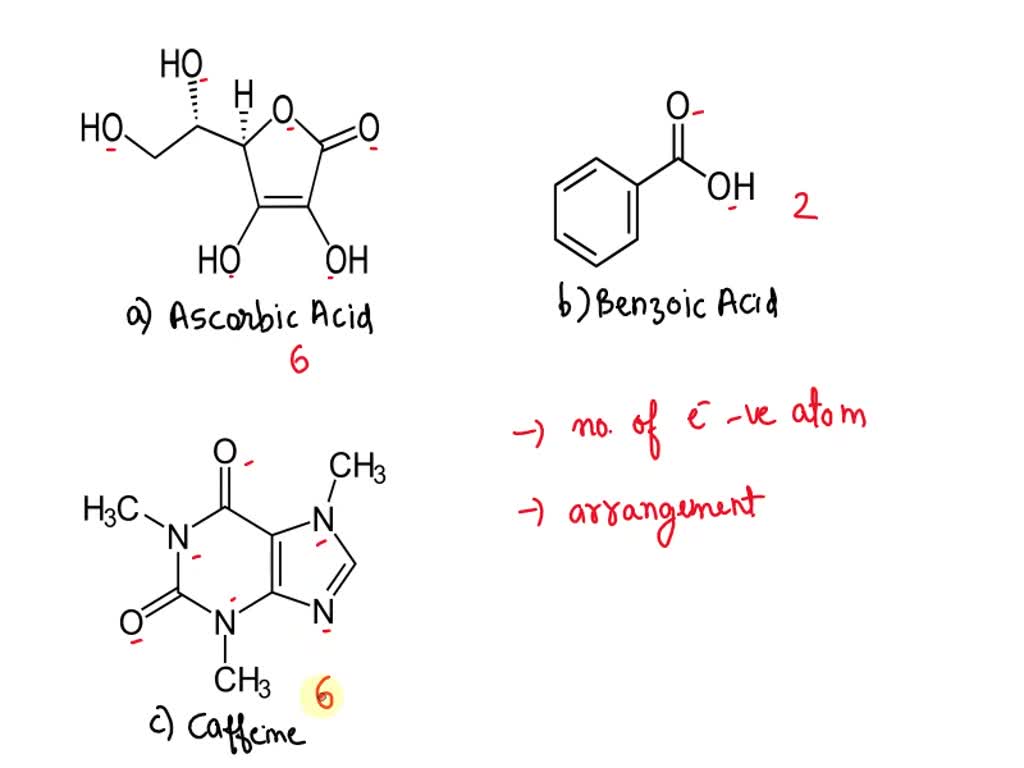
SOLVED: Please rank the following molecules from most polar to least polar. a) ascorbic acid b) benzoic acid c) caffeine

How can benzoic acid dissolve into diethyl ether since benzoic acid is polar and diethyl ether is non-polar? | Homework.Study.com

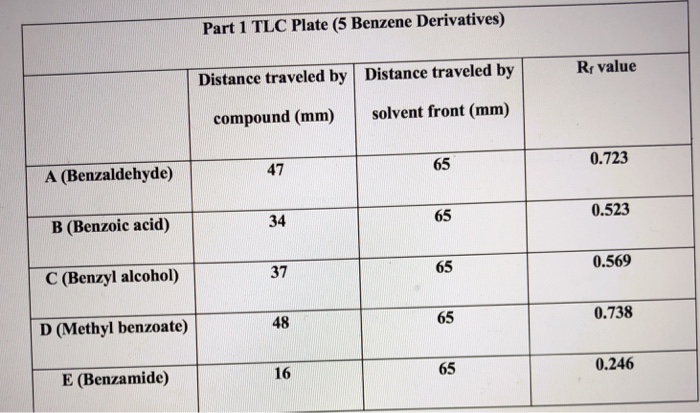
SOLVED: You were given samples of toluene, benzyl alcohol, benzoic acid and benzaldehyde. You spotted these 4 compounds on a silica gel TLC plate and run that plate in dichloromethane solvent. Predict

1) Rank trans-stilbene, benzoic acid, and fluorenone by polarity (least polar to most polar). Provide an explanation for your proposed ranking. (2) Hexane, ethyl acetate and acetone will be used as

Measured 20 log P values for benzoic acid and aniline derivatives of... | Download Scientific Diagram

2. Draw the chemical structure of benzoic acid and use it to answer the following solubility-based questions: a) Circle and label the region of the molecule that exhibits non-polar behavior. b) Thes